One-on-one Wellness Guidance
You’re Not Like Everybody Else. So Why Should Your Health Solutions Be?
Our Compounding Pharmacy Is Right For You If:
- Medications no longer provide the relief they used to.
- You’re worried you’re overmedicating.
- You feel helpless when medications, supplements, and hormone treatments don’t work.
- You’re experiencing a strain on relationships due to hormone imbalances.

Get Your Life Back
Triangle Compounding Pharmacy Provides Personalized Service To Support Your Body With The Unique Solutions It Deserves.
Women’s Health
Most hormone replacement treatments (hrt) are synthetic and available in very limited doses, which can cause intolerable side effects. Compounded hrt provides individualized doses and follows the same metabolic pathway as the body’s own hormones.
Men’s Health
As you age, men often experience issues like decreased libido, erectile dysfunction, and depression from imbalanced hormones. Our experienced staff will discreetly discuss your needs and help you get back to your best.
Allergies & Sensitivities
Get personalized prescription medication without non-essential ingredients or contaminants, such as gluten, dye, lactose, etc. to which a patient may be allergic. Sensitivities and allergies shouldn’t keep you from getting the medication you need!
Wellness Solutions
Optimizing Health Requires The Right Combination Of Support. Triangle Compounding Pharmacy Offers A Variety Of Premium Over The Counter Solutions To Support Your Health Needs.
People Are Our Priority
Your Body Is Unique. You Deserve Medication And Supplements As Specialized As You. Check Out How We’Ve Helped Our Customers.
Our Process to Personalize Your Prescriptions
Schedule A Free Consultation
We want to ensure we are the best fit for you. That’s why every FREE consultation includes a custom health assessment.
Get Your Personalized Medication
Every BODY is unique. We create individualized solutions for you – not “everybody.”
Live At Your Best
You deserve to live your life how you want! Get the solutions to tackle your unique health challenges and feel like yourself again.

Life At Your Best Is
Possible
Possible
It’s tough when you or a loved one don’t feel your best. You feel like no one is listening, you’re running out of options, and don’t know how to start feeling better.
It’s negatively impacting your life and you’re worried about what this means for your future.
At Triangle Compounding Pharmacy, we specialize in creating personalized compounded medications that are optimized specifically for your unique medical needs.
You shouldn’t have to suffer when there’s a solution – made just for you. Equip your body so that you can feel your best as soon as possible.

Shop Our Online Store for Professional-Grade
Supplements
Supplements
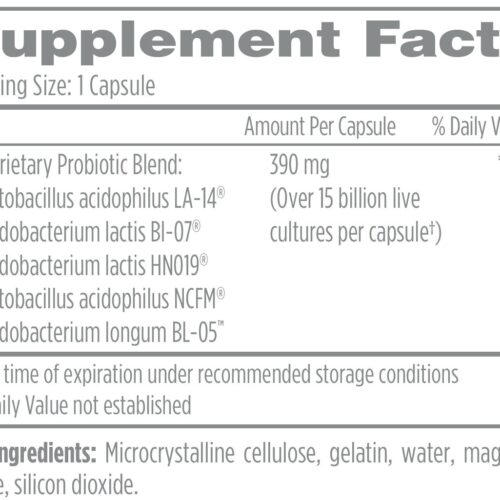
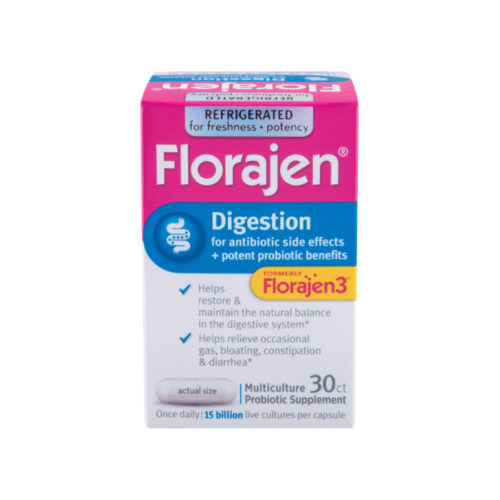
You’re looking for high quality supplements, but you don’t know where to find them. The truth is that most supplements found in big chain retail or online stores sell supplements made with low-quality ingredients and fillers.
Triangle Compounding Pharmacy offers only the highest quality professional-grade supplements available. We’ve hand-selected each product to ensure that it meets our rigorous standards for quality and efficacy.
It’s easy for you to order your favorite supplements directly from us.
Persistent Health Challenges Demand The Best Service
Our team has 60+ years of combined experience in compounding pharmacy. We own and operate from a state-of-the-art facility where we train colleagues and others in our field. We’ll go to any length necessary to get you the quality and custom health solutions you deserve.

Is Hormone Optimization Right For Me?
Error: Contact form not found.